How Much Blood Volume Does Plasma Makeup

Blood plasma is a light amber-colored liquid component of blood in which blood cells are absent-minded, just contains proteins and other constituents of whole claret in suspension. It makes up about 55% of the torso's total claret book.[1] It is the intravascular role of extracellular fluid (all torso fluid outside cells). Information technology is mostly water (upwardly to 95% by book), and contains important dissolved proteins (6–8%; e.thousand., serum albumins, globulins, and fibrinogen),[two] glucose, clotting factors, electrolytes (Na +
, Ca ii+
, Mg two+
, HCO3 - , Cl −
, etc.), hormones, carbon dioxide (plasma existence the main medium for excretory product transportation), and oxygen. It plays a vital role in an intravascular osmotic effect that keeps electrolyte concentration balanced and protects the body from infection and other blood-related disorders.[3]
Blood plasma is separated from the blood by spinning a vessel of fresh blood containing an anticoagulant in a centrifuge until the claret cells fall to the bottom of the tube. The blood plasma is then poured or fatigued off.[4] For point-of-intendance testing applications, plasma can be extracted from whole blood via filtration[5] or via agglutination[6] to allow for rapid testing of specific biomarkers. Claret plasma has a density of approximately 1,025 kg/yard3 (one.025 g/ml).[7]
Blood serum is blood plasma without clotting factors.[4]
Plasmapheresis is a medical therapy that involves blood plasma extraction, treatment, and reintegration.
Fresh frozen plasma is on the WHO Model List of Essential Medicines, the most important medications needed in a basic health system.[8] It is of critical importance in the treatment of many types of trauma which outcome in blood loss, and is therefore kept stocked universally in all medical facilities capable of treating trauma (e.g., trauma centers, hospitals, and ambulances) or that pose a take chances of patient blood loss such equally surgical suite facilities.
Volume [edit]
![]()
![]()
The same information, shown in molarity rather than mass.
Claret plasma volume may be expanded by or drained to extravascular fluid when there are changes in Starling forces across capillary walls. For example, when claret pressure drops in circulatory shock, Starling forces drive fluid into the interstitium, causing 3rd spacing.[ citation needed ]
Standing still for a prolonged period will cause an increase in transcapillary hydrostatic pressure. Equally a result, approximately 12% of blood plasma volume will cross into the extravascular compartment. This causes an increase in hematocrit, serum total protein, blood viscosity and, as a upshot of increased concentration of coagulation factors, it causes orthostatic hypercoagulability.[9]
Plasma proteins [edit]
Albumins [edit]
Serum albumins are the most common plasma proteins and they are responsible for maintaining the osmotic pressure of blood. Without albumins, the consistency of claret would exist closer to that of water. The increased viscosity of claret prevents fluid from inbound the bloodstream from outside the capillaries. Albumins are produced in the liver bold the absenteeism of a hepatocellular deficiency.[10]
Globulins [edit]
The second most common type of poly peptide in the claret plasma are globulins. Important globulins include immunoglobins which are important for the allowed system and send hormones and other compounds around the body. There are iii main types of globulins. Alpha-1 and Alpha-2 globulins are formed in the liver and play an important role in mineral transport and the inhibition of blood coagulation.[11] An example of beta globulin found in blood plasma includes low-density lipoproteins (LDL) which are responsible for transporting fat to the cells for steroid and membrane synthesis.[12] Gamma globulin, better known every bit immunoglobulins, are produced past plasma B cells, and provides the human torso with a defence force system confronting invading pathogens and other allowed diseases.[xiii]
Fibrinogen [edit]
Fibrinogen proteins make up well-nigh of the remaining proteins in the claret. Fibrinogens are responsible for clotting blood to help preclude blood loss.[fourteen]
Color [edit]

Plasma is normally yellow due to bilirubin, carotenoids, hemoglobin and transferrin.[15] In abnormal cases, plasma tin can have varying shades of orangish, greenish or brown. Green color tin be due to ceruloplasmin or sulfhemoglobin. Latter may form due to medicines that are able to form sulfonamides once ingested (see sulfhemoglobinemia).[16] Dark brown or blood-red colour can appear due to hemolysis, in which methemoglobin is released from broken blood cells (run across methemoglobinemia).[17] Plasma is unremarkably relatively transparent, but sometimes it can be opaque. Opaqueness is typically due to elevated content of lipids similar cholesterol and triglycerides (see hyperlipidemia).[18]
Plasma vs. serum in medical diagnostics [edit]
Blood plasma and claret serum are ofttimes used in blood tests. Some tests tin can be washed only on plasma and some only on serum. Some can exist washed on both, only depending on the test, use of either plasma or serum can be more practical.[19] In addition, some tests take to exist done with whole claret, such as the determination of the amount of claret cells in claret via flow cytometry.[twenty]
| Some of the benefits of plasma over serum | Some of the benefits of serum over plasma |
|---|---|
| Plasma preparation is quick, as it is not coagulated. Serum sample preparation requires almost 30 minutes of waiting time earlier information technology can be centrifuged and then analyzed.[19] However, coagulation tin be hastened down to a few minutes by calculation thrombin or similar agents to the serum sample.[21] | Plasma preparation requires the add-on of anticoagulants, which can cause expected and unexpected measurement errors. For example, anticoagulant salts can add together extra cations similar NHfour +, Li+, Na+ and K+ to the sample,[19] or impurities like lead and aluminum.[22] Chelator anticoagulants like EDTA and citrate salts work past bounden calcium (see carboxyglutamic acrid), but they may also demark other ions. Even if such ions are not the analytes, chelators tin interfere with enzyme action measurements. For case, EDTA binds zinc ions, which alkaline phosphatases need as cofactors. Thus, phosphatase activity cannot be measured if EDTA is used.[19] |
| Compared to serum, fifteen–20% larger volume of plasma can exist obtained from a blood sample of certain size. Serum lacks some proteins that partake in coagulation and increase the sample volume.[xix] | An unknown book of anticoagulants can exist added to a plasma sample past accident, which may ruin the sample as the analyte concentration is changed past an unknown amount.[22] |
| Serum preparation can cause measurement errors past increasing or decreasing the concentration of the analyte that is meant to be measured. For example, during coagulation, claret cells consume blood glucose and platelets increase the sample content of compounds similar potassium, phosphates and aspartate transaminase past secreting them. Glucose or these other compounds may be the analytes.[19] | No anticoagulants are added to serum samples, which decreases the grooming cost of the samples relative to plasma samples.[22] |
| Plasma samples can form tiny clots if the added anticoagulant is not properly mixed with the sample. Non-uniform samples tin cause measurement errors.[22] |
History [edit]

Private Roy West. Humphrey is beingness given blood plasma later on he was wounded by shrapnel in Sicily in August 1943.
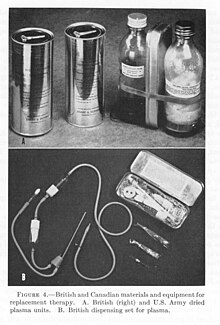
Dried plasma packages used by the British and US militaries during WWII.
Plasma was already well known when described by William Harvey in de Mortu Cordis in 1628, simply knowledge of it probably extends as far dorsum every bit Vesalius (1514–1564). The discovery of fibrinogen past William Henson, c. 1770,[23] made it easier to written report plasma, as ordinarily, upon coming in contact with a strange surface – something other than vascular endothelium – clotting factors become activated and clotting proceeds quickly, trapping RBCs etc. in the plasma and preventing separation of plasma from the claret. Adding citrate and other anticoagulants is a relatively recent advance. Note that, upon formation of a clot, the remaining clear fluid (if whatsoever) is Serum (claret), which is essentially plasma without the clotting factors.[ citation needed ]
The use of claret plasma every bit a substitute for whole blood and for transfusion purposes was proposed in March 1918, in the correspondence columns of the British Medical Journal, past Gordon R. Ward. "Dried plasmas" in powder or strips of material format were developed and first used in World War Ii. Prior to the United states' involvement in the state of war, liquid plasma and whole blood were used.[ citation needed ]
The origin of plasmapheresis [edit]
Dr. José Antonio Grifols Lucas, a scientist from Vilanova i la Geltrú, Spain,[24] founded Laboratorios Grifols in 1940.[25] Dr. Grifols pioneered a commencement-of-its-kind technique called plasmapheresis,[25] where a donor'south red claret cells would be returned to the donor'due south body almost immediately after the separation of the blood plasma. This technique is all the same in do today, almost 80 years after. In 1945, Dr. Grifols opened the world's first plasma donation center.[24] Thirteen years after the middle's opening, Dr. Grifols unexpectedly died at the young age of 41 due to leukemia.
Blood for Britain [edit]
The "Claret for Britain" plan during the early 1940s was quite successful (and popular in the United States) based on Charles Drew's contribution. A large project began in Baronial 1940 to collect blood in New York City hospitals for the consign of plasma to Britain. Drew was appointed medical supervisor of the "Plasma for Britain" project. His notable contribution at this fourth dimension was to transform the test tube methods of many blood researchers into the first successful mass product techniques.[ commendation needed ]
Nonetheless, the decision was made to develop a dried plasma package for the armed forces as information technology would reduce breakage and make the transportation, packaging, and storage much simpler.[26] The resulting dried plasma package came in two tin cans containing 400 cc bottles. One bottle independent plenty distilled water to reconstitute the dried plasma independent within the other bottle. In most 3 minutes, the plasma would be fix to utilize and could stay fresh for around four hours.[26] The Blood for Great britain program operated successfully for 5 months, with total collections of almost 15,000 people donating blood, and with over 5,500 vials of blood plasma.[27]
Post-obit the Supplying Blood Plasma to England project, Drew was named manager of the Reddish Cross blood bank and banana director of the National Enquiry Quango, in charge of blood collection for the United States Army and Navy. Drew argued confronting the armed forces directive that blood/plasma was to be separated past the race of the donor. Drew insisted that at that place was no racial departure in human blood and that the policy would lead to needless deaths as soldiers and sailors were required to wait for "same race" blood.[28]
By the end of the war the American Red Cross had provided enough claret for over six million plasma packages. Almost of the surplus plasma was returned to the United States for civilian use. Serum albumin replaced dried plasma for gainsay utilize during the Korean State of war.[26]
Plasma donation [edit]
Plasma as a claret product prepared from blood donations is used in claret transfusions, typically as fresh frozen plasma (FFP) or Plasma Frozen inside 24 hours after phlebotomy (PF24). When donating whole blood or packed ruby blood cell (PRBC) transfusions, O- is the most desirable and is considered a "universal donor," since it has neither A nor B antigens and can be safely transfused to near recipients. Type AB+ is the "universal recipient" type for PRBC donations. However, for plasma the situation is somewhat reversed. Blood donation centers will sometimes collect only plasma from AB donors through apheresis, equally their plasma does not contain the antibodies that may cross react with recipient antigens. As such, AB is oftentimes considered the "universal donor" for plasma. Special programs be merely to cater to the male person AB plasma donor, considering of concerns about transfusion related astute lung injury (TRALI) and female donors who may accept college leukocyte antibodies.[29] Still, some studies evidence an increased risk of TRALI despite increased leukocyte antibodies in women who have been significant.[thirty]
United Kingdom [edit]
Following fears of variant Creutzfeldt-Jakob illness (vCJD) being spread through the claret supply, the British government began to phase out blood plasma from U.Chiliad. donors and past the end of 1999 had imported all claret products made with plasma from the Usa.[31] In 2002, the British regime purchased Life Resources Incorporated, an American blood supply visitor, to import plasma.[32] The company became Plasma Resource UK (PRUK) which owned Bio Products Laboratory. In 2013, the British government sold an 80% pale in PRUK to American hedge fund Bain Uppercase, in a bargain estimated to exist worth £200 million. The auction was met with criticism in the UK.[33] In 2009, the U.K. stopped importing plasma from the United States, as it was no longer a viable pick due to regulatory and jurisdictional challenges.[34]
Currently,[ when? ] blood donated in the United Kingdom is used by United kingdom Blood Services for the industry of plasma blood components (Fresh Frozen Plasma (FFP) and cryoprecipitate). However, plasma from United kingdom donors is all the same not used for the commercial manufacture of fractionated plasma medicines.[ citation needed ]
Synthetic blood plasma [edit]
Simulated trunk fluid (SBF) is a solution having a similar ion concentration to that of human blood plasma. SBF is normally used for the surface modification of metallic implants, and more recently in gene commitment application.
See also [edit]
- Blood plasma fractionation
- Blood proteins
- Chromatography in claret processing
- Diag Man
- Hypoxia preconditioned plasma
- Intravascular volume status
References [edit]
- ^ Dennis O'Neil (1999). "Blood Components". Palomar Higher. Archived from the original on June 5, 2013.
- ^ Tuskegee University (May 29, 2013). "Chapter 9 Blood". tuskegee.edu. Archived from the original on December 28, 2013.
- ^ "Means to Go along Your Claret Plasma Healthy". Archived from the original on November i, 2013. Retrieved November ten, 2011.
- ^ a b Maton, Anthea; Jean Hopkins; Charles William McLaughlin; Susan Johnson; Maryanna Quon Warner; David LaHart; Jill D. Wright (1993). Human Biology and Health . Englewood Cliffs, New Bailiwick of jersey, United states of america: Prentice Hall. ISBN0-13-981176-ane.
- ^ Tripathi South, Kumar V, Prabhakar A, Joshi South, Agrawal A (2015). "Passive blood plasma separation at the microscale: a review of blueprint principles and microdevices". J. Micromech. Microeng. 25 (viii): 083001. Bibcode:2015JMiMi..25h3001T. doi:10.1088/0960-1317/25/8/083001.
- ^ Guo, Weijin; Hansson, Jonas; van der Wijngaart, Wouter (2020). "Constructed Paper Separates Plasma from Whole Blood with Low Protein Loss". Belittling Chemistry. 92 (nine): 6194–6199. doi:10.1021/acs.analchem.0c01474. ISSN 0003-2700. PMID 32323979.
- ^ Shmukler, Michael (2004). Elert, Glenn (ed.). "Density of blood". The Physics Factbook. Archived from the original on December ix, 2021. Retrieved Jan 23, 2022.
- ^ "19th WHO Model List of Essential Medicines (April 2015)" (PDF). WHO. April 2015. Archived (PDF) from the original on April 28, 2019. Retrieved May 10, 2015.
- ^ Masoud M, Sarig Yard, Brenner B, Jacob Thousand (June 2008). "Orthostatic hypercoagulability: a novel physiological mechanism to activate the coagulation system". Hypertension. 51 (six): 1545–51. doi:x.1161/HYPERTENSIONAHA.108.112003. PMID 18413485.
- ^ "Albumin: Liver Role Test - Viral Hepatitis and Liver Disease". Archived from the original on September 13, 2021. Retrieved March 15, 2021.
- ^ "Globulins | Encyclopedia.com". www.encyclopedia.com. Archived from the original on November 29, 2021. Retrieved Nov 29, 2021.
- ^ "Blood Plasma Components and Role". News-Medical.net. October 10, 2018. Archived from the original on November 29, 2021. Retrieved November 29, 2021.
- ^ Biga, Lindsay M.; Dawson, Sierra; Harwell, Amy; Hopkins, Robin; Kaufmann, Joel; LeMaster, Mike; Matern, Philip; Morrison-Graham, Katie; Quick, Devon; Runyeon, Jon (September 26, 2019). "xviii.ane Functions of Blood". Archived from the original on November 29, 2021. Retrieved Nov 29, 2021.
- ^ Basic Biological science (2015). "Claret cells". Archived from the original on July xviii, 2021. Retrieved March 17, 2020.
- ^ Elkassabany NM, Meny GM, Doria RR, Marcucci C (2008). "Dark-green Plasma—Revisited". Anesthesiology. 108 (iv): 764–765. doi:ten.1097/ALN.0b013e3181672668. PMID 18362615. Archived from the original on July 27, 2020. Retrieved March 21, 2020.
- ^ Mani A, Poornima AP, Gupta D (2019). "Greenish discoloration of plasma: Is information technology really a affair of concern?". Asian Journal of Transfusion Science. 13 (1): one–2. doi:10.4103/ajts.AJTS_117_18. PMC6580839. PMID 31360002.
- ^ Tesfazghi MT, McGill MR, Yarbrough ML (2019). "What's Causing This Dark Brown Plasma?". The Journal of Applied Laboratory Medicine. iv (1): 125–129. doi:ten.1373/jalm.2018.026633. PMID 31639715.
- ^ Agnihotri N, Kumar 50 (2014). "Turbid plasma donations: Demand for quantification". Asian Periodical of Transfusion Science. 8 (2): 78–79. doi:ten.4103/0973-6247.137436. PMC4140067. PMID 25161342.
- ^ a b c d east f "Use of anticoagulants in diagnostic laboratory investigations". Earth Health Organization. 2002. hdl:10665/65957. WHO/DIL/LAB/99.1 Rev.2.
- ^ Jimenez VE, Chew Y, Nicholson L, Burns H, Anderson P, Chen H, Williams L, Keung K, Zanjani NT, Dervish S, Patrick E (2019). "Standardisation of menstruation cytometry for whole claret immunophenotyping of islet transplant and transplant clinical trial recipients". PLOS ONE. 14 (five): e0217163. Bibcode:2019PLoSO..1417163J. doi:10.1371/journal.pone.0217163. PMC6530858. PMID 31116766.
- ^ Kocijancic 1000, Cargonja J, Delic-Knezevic A (2014). "Evaluation of the BD Vacutainer® RST claret collection tube for routine chemistry analytes: clinical significance of differences and stability study". Biochemia Medica. 24 (3): 368–375. doi:x.11613/BM.2014.039. PMC4210257. PMID 25351355.
- ^ a b c d Uges D (1988). "Plasma or serum in therapeutic drug monitoring and clinical toxicology". Pharmaceutisch Weekblad. x (5): 185–188. doi:ten.1007/BF01956868. PMID 3060834. S2CID 32330414.
- ^ Wintrobe. Blood, Pure and Eloquent.
- ^ a b "When a Dream Comes True". grifols.com. Jan 2015. Archived from the original on July 25, 2021. Retrieved March 21, 2020.
- ^ a b "Biography: J.A. Grifols". discovertheplasma.com. Archived from the original on July 25, 2021. Retrieved March 21, 2020.
- ^ a b c "Abode". achh.army.mil. Archived from the original on Dec xi, 2021. Retrieved December 9, 2021.
- ^ Starr, Douglas P. (2000). Blood: An Ballsy History of Medicine and Commerce. New York: Quill. ISBN0-688-17649-six.
- ^ Hirsch, Eric (1991). What Your 1st Grader Needs to Know: Fundamentals of a Expert Offset-Grade Teaching . pp 232–233. New York: Doubleday. ISBN9780385411172.
- ^ "AB Plasma Donor Program". NIH Clinical Center. March 20, 2008. Archived from the original on February 15, 2022. Retrieved March 18, 2011.
- ^ "Female Plasma May Not Increase Risk for Transfusion-Related Acute Lung Injury". Medscape. October 23, 2007. Archived from the original on February 12, 2022. Retrieved July ii, 2011.
- ^ Roos, Robert. "Claret transmission of vCJD suspected in Britain". Center for Infectious disease Inquiry and Policy. Archived from the original on June 24, 2021. Retrieved June 24, 2021.
- ^ "NHS pays £50m for The states claret plasma house". The Guardian. December 17, 2002. Archived from the original on June 24, 2021. Retrieved June 24, 2021.
- ^ Rankin, Jennifer (July 18, 2013). "Bain Uppercase buys bulk stake in Plasma Resources UK". The Guardian. Archived from the original on November 13, 2020. Retrieved June 24, 2021.
- ^ "Importation of plasma and use of apheresis platelets as take chances reduction measures for variant Creutzfeldt-Jakob Affliction" (PDF). Archived (PDF) from the original on July 25, 2021. Retrieved June 24, 2021.
How Much Blood Volume Does Plasma Makeup,
Source: https://en.wikipedia.org/wiki/Blood_plasma
Posted by: smithoneten.blogspot.com


0 Response to "How Much Blood Volume Does Plasma Makeup"
Post a Comment